(Reuters) -Regeneron Pharmaceuticals said on Monday the U.S. FDA has declined approval for its blood cancer therapy for two forms of lymphoma, raising concerns over the progress of ongoing confirmatory trials.
In its so-called complete response letter (CRL), the U.S. Food and Drug Administration said it needed more data from enrollments in dose-finding and confirmatory portions of trials, delaying its decision on the drug.
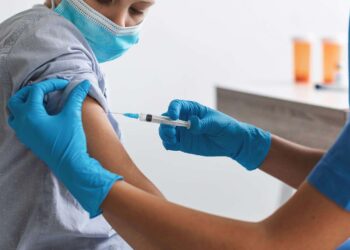
The company was testing its experimental drug, odronextamab, in multiple late-stage trials in patients with Follicular lymphoma and Diffuse large B-cell lymphoma, the two most common subtypes of non-Hodgkin lymphoma.
“To our knowledge, this is the first time the FDA has issued a CRL for this reason,” a Regeneron spokesperson said about the decision by the FDA, which did not identify any issues with the drug’s clinical efficacy or safety, trial design, labeling or manufacturing.
Shares of the company fell nearly 2% to $948.90 in premarket trading. The company was looking to expand its oncology portfolio with odronextamab, beyond its lone approved cancer drug, Libtayo.
“While we acknowledge the general concerns that FDA has about sponsors failing to complete their post-marketing confirmatory trials, the relevant laws and regulatory guidances do not lay out rigid criteria for assessing whether the progress on a confirmatory trial is adequate to allow for an accelerated approval,” the Regeneron spokesperson said.
(Reporting by Sneha S K, Puyaan Singh and Sriparna Roy in Bengaluru; Editing by Shinjini Ganguli)
Source link : https://www.medscape.com/s/viewarticle/us-fda-declines-approve-regenerons-blood-cancer-therapy-2024a10005ly?src=rss
Author :
Publish date : 2024-03-25 11:29:54
Copyright for syndicated content belongs to the linked Source.